Local mixed forest habitat, showing the grandeur of winter collecting. Photo taken 16 January 2023.
Continuing my trend from 2021 and 2022, it’s time for the summary of my myriapod collecting during 2023! I made a concerted effort last year to collect less so that I could focus on other things (mainly writing, curation of my specimens, and job hunting), but I still found a number of interesting and rare species. An added bonus from last year is that a number of friends and colleagues sent me specimens from far-flung areas, which added some exceptional species to my collection. I want to give a particular thank you to Lance Andrew, Joe Girgente, Adam Haberski, Curt Harden, Brendan O'Loughlin, and Carol Tingley for sending me their specimens.
Thanks to the specimens sent to me by colleagues, the collecting map spans the continent! The Alaska locality especially sticks out, and represents some beautiful Parajulidae.
Map of 2023 collecting localities
I totaled 37 collecting events in 2023, spanning only two states: Virginia and Ohio. All the dots on the map from outside those states are specimens sent to me by other people, and those specimens really augmented my collection last year! The numbers I’m using for the rest of this post include the specimens sent by colleagues.
Total myriapod specimens collected in 2023
All in all, my colleagues and I collected 1,450 myriapods in 2023, as shown in the above graph (blue bars, left axis). The total number of collection events are represented by the orange line, with numbers reflected on the right axis. There were a total of 81 collection events last year: I did 37 of them, while my colleagues conducted 44. Again, the value of others collecting myriapods for me is immense!
These collections included 109 species from 36 families, beating 2022’s count of 88 species from 30 families. Those 109 species include 71 genera, including a number of unique genera I didn’t have in my collection before. Last year I wrote about wanting to top 100 species in a year, and I achieved that goal!
Since this is my third year of tracking this data, I can include my previous years of data to see how my collecting habits have differed.
Total specimens collected monthly, 2021-2023
The above graph shows the total number of specimens collected during each month from 2021-2023. There’s quite a bit of variation there, mostly reflecting collecting effort. The totals from 2021 are quite low until September 2021, since that’s when I started using my homemade Berlese funnels. The main takeaway here is that collection effort is typically the most important determiner of how many specimens I collect each month, with local weather playing a part as well. Totals from November through February tend to be lower than Spring through Fall, but there are, of course, exceptions. Additionally, if you want to get the full picture of your local myriapod diversity, you’ll miss unique cold weather-active species if you don’t collect between November and February.
I’ve collected throughout the year enough now to recognize a few distinct myriapod faunal groups. There are species like Nadabius pullus (Lithobiomorpha: Lithobiidae) that you can find easily throughout the year. Other groups like the Apheloriini (Polydesmida: Xystodesmidae) are most active during the spring and fall. And then there are other species, like the genus Garibius (Lithobiomorpha: Lithobiidae) and many Chordeumatida, which are active during late fall through early spring. Figuring out which species fit these various seasonal patterns (and which ones don’t!) is one of the most fulfilling parts of collecting myriapods for me personally. The cold weather-active species in particular are some of the most exciting to find, as these are typically rare in collections, despite their ubiquity during their active season. Besides, you pretty much have the entire woods to yourself while searching for them. It’s quite pleasant to grub around in the dirt in brisk weather.
Another fun thing to check the yearly data for is the number of unique species per month, shown in the graph below:
Number of species collected monthly, 2021-2023
These monthly totals are somewhat less variable than the total specimens collected each month, but collecting effort still has a strong influence (see November 2023!). These totals can be highly influenced by colleagues sending me specimens, particularly if they’re based outside of Virginia. The standout from this chart is April 2023, with the most species I’ve ever collected in a month, at 51! That was also the month with the highest total specimens, at 297 (see previous graph).
Diving into the 2023 species-specific data, there are similar trends to my 2022 collecting. Once again, the centipede Nadabius pullus (Lithobiomorpha: Lithobiidae) was my most-collected species, represented by 154 specimens, less than last year’s count of 195 specimens. The full top ten list is:
Nadabius pullus (154), Nampabius sp 1 (90), Arctogeophilus umbraticus (70), Oxidus gracilis (70), Hanseniella sp (63), Strigamia branneri (60), Typhlobius sp (52), Ptyoiulus impressus (49), Chaetaspis sp (40), Paitobius zinus (36)
The four names in bold are species that weren’t on last year’s list. Collectively, these 10 species account for 51% of all myriapods collected in 2023. At the other end of the spectrum, 32 species are represented by 1 or 2 specimens, making up only 3% of the total specimens collected, but 29% of the total species collected. Sometimes those singletons pack a big punch diversity-wise.
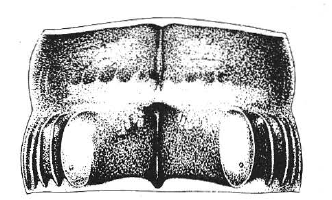
I was able to find a number of lifer species in 2023, including some I had been after for years! One of my favorites was the centipede Agathothus gracilis (Geophilidae: Linotaeniinae), which has only been reported a few times from Tennessee and southwest Virginia. It’s one of only two genera of Linotaeniinae in eastern North America, and is far rarer than the common Strigamia. I wasn’t sure I’d be able to recognize a specimen of A. gracilis, but after seeing the forcipules, it’s unmistakeable—they’re flattened and sharp, and lack the tarsungular tooth of Strigamia, making A. gracilis immediately recognizable.
Agathothus gracilis, head and anterior segments, ventral view.
By far my most surprising find of 2023 was a liliputian geophilomorph, the enigmatic Nannarrup hoffmani. Funnily enough, 2023 marks the species’ 20th birthday, having originally been described from Central Park in New York City in 2003. I found half a dozen specimens in southwest Virginia, while looking for a different species entirely! The species itself is native to an unknown part of east Asia, and only recently has the genus been found in its native range (see Tsukamoto et al. 2022). This is a new state record for the species (and the family) and makes it likely that N. hoffmani will turn up in additional states, if searched for.
Nannarrup hoffmani: live specimen in soil matrix (left) and ventral view of forcipules (right).
Another geophilomorph I was happy to find was Strigamia hoffmani, a species only described in 2009 in honor of Richard Hoffman’s 80th birthday. It’s a small species that can be difficult to separate from Virginia’s other species of Strigamia, and is only known from Tazewell and Roanoke counties. In 2023, I found it in two additional counties: Grayson and Giles. The key to finding this one may be searching areas over 1,000 m in elevation, suggesting a more boreal-adapted species. It still hasn’t been found outside of Virginia, so get searching, those of you in surrounding states!
Last year brought me my first specimen of Lamyctes pius (Lithobiomorpha: Henicopidae), likely a synonym of L. emarginatus (see Shear 2018). Chamberlin described a number of species within the genus Lamyctes, but the evidence points to them being an introduced species in North America. Native henicopids are rare here, as the family is mainly confined to the southern hemisphere.
Lamyctes pius (Lithobiomorpha: Henicopidae) head, dorsal view (left) and ventral view (right)
Moving on to the millipedes, there were a number of surprises this year. Down in Wise County, I found my first sphaeriodesmid in Virginia: Desmonus earlei!
Desmonus earlei, lateral view
This is a slight range extension and only the second record for Virginia that I know of; previously it had been recorded from Lee County. There’s another Desmonus species that ranges from Missouri south to Texas and adjacent Mexico, Desmonus pudicus. Both species are quite small, only about 5 mm across when curled into the loose spiral pictured above, and maybe a centimeter fully extended. Despite their small size, they’re immediately recognizable by the large dorsal tubercles on their rings, and by their coating of dirt. While Desmonus is the only representative that occurs in the US, the family Sphaeriodesmidae is more diverse in Mexico and down through South America, and it’s always a treat to see one of these species.
Desmonus earlei gonopods, posterior view (left) and lateral view (right)
There’s one more polydesmidan millipede I want to highlight, and it was by far the biggest millipede find of my year. I was meeting a friend at a park in Wythe County, and while I was waiting, I scratched around in the leaf litter near a stream to see what I could find. Within five minutes, I found some small cherry millipedes (Xystodesmidae) and did a double take:
Rhysodesmus restans, dorsal view of live specimen
“No way,” I whispered to myself. This was Rhysodesmus restans, something of an artifact in Appalachia. Almost all species of Rhysodesmus are further south, in Texas, Mexico, and Central America. But there are a few still around, left behind in the Appalachian Mountains. I’d been searching for this species for years at this point, and always much further away, down in Washington County (the type locality). Even better: they were abundant here! I collected a number of specimens and left more behind, still in disbelief at my luck.
The final species I’ll highlight is a hidden gem that I’d actually collected multiple times in the past few years, but which I had misidentified. This was pointed out to me by my colleague Pierre-Marc Brousseau, to whom I’m grateful! A small millipede in the order Julida, the species Okeanobates americanus is one I had read about, but that was the limit of my experience with it.
Okeanobates americanus, lateral view
It turns out that I had unknowingly collected this species almost yearly since 2020! I had it misidentified as an unknown species of Blaniulidae, but Pierre-Marc noticed the rectangular eye patch in one of my iNaturalist photos and corrected me. The species ranges from North Carolina up through Quebec, Canada, but is uncommon. It seems to favor life within logs, as I’ve never found it in leaf litter. I included more information about the species and the Okeanobatidae in a blog post back in November, so check that out to learn more. It’s not often that I get to add a new family to my collection, what a thrill!
That about wraps up my 2023 review. I found some superb species last year, and much of that was thanks to friends and colleagues, to whom I’m indebted for sending me specimens.
Possibly the strangest thing I did last year was that I accidentally ended up writing a key to all eastern geophilomorph centipedes. I meant to focus on Lithobiomorpha in 2023, but got hooked on those weird little soil centipedes. If you’re interested in using the key or sending me geophilomorphs from your region, please get in touch. It will be a little while before I publish it, but I’m aiming to write up centipede resources in the next few years to focus attention on the North American fauna, especially in the east.
This post has gone on long enough, so I’ll end it here with a photo comparison of the new litter sifter I bought in 2023. It replaces my previous sifter, which I’d been using for almost a decade. Can you tell which is which?
Further reading:
Chamberlin RV (1912) The Geophiloidea of the southeastern states. Bulletin of the Museum of Comparative Zoology at Harvard College 54: 405–436. https://www.biodiversitylibrary.org/page/2815547
Enghoff H (1979) The millipede genus Okeanobates (Diplopoda, Julida: Nemasomatidae). Steenstrupia 5: 161–178.
Hoffman RL (1998) An Appalachian species of Rhysodesmus (Polydesmida: Xystodesmidae: Rhysodesmini). Myriapodologica 5: 77–83. https://www.biodiversitylibrary.org/page/52243216
Pereira LA (2009) A new dwarf species of the genus Strigamia Gray, 1843 from the southern Appalachian Mountains of western Virginia (Chilopoda: Geophilomorpha: Linotaeniidae). Virginia Museum of Natural History Special Publication 16: 209–222.
Shear WA (2018) The centipede family Anopsobiidae new to North America, with the description of a new genus and species and notes on the Henicopidae of North America and the Anopsobiidae of the Northern Hemisphere (Chilopoda, Lithobiomorpha). Zootaxa 4422: 259–283. https://doi.org/10.11646/zootaxa.4422.2.6
Shelley RM (2000) Revision of the milliped subfamily Desmoninae (Polydesmida: Spheriodesmidae). Myriapodologica 6: 27–54. https://www.vmnh.net/content/vmnh/uploads/PDFs/research_and_collections/myriapodologica/myriapodologica_v6_5.pdf
Tsukamoto S, Shimano S, Eguchi K (2022) Two new species of the dwarf centipede genus Nannarrup Foddai, Bonato, Pereira & Minelli, 2003 (Chilopoda, Geophilomorpha, Mecistocephalidae) from Japan. ZooKeys 1115: 117–150. https://doi.org/10.3897/zookeys.1115.83946